|
Perhaps due to its unique role, nucleoplasmin's similarity to a group of proteins implicated in chromatin regulation, such as the plant type 2 histone deacetylases, or HD-tuins, and the yeast Fpr3 and Fpr4 proteins, has never been noticed. An electron microscopy study of native hyper-phosphorylated NUP concluded that five H2A/H2B dimers bind one NUP core particle or one histone dimer per NUP subunit. Its structure was solved revealing a homo-pentamer, prompting the question of how a molecule with 5-fold symmetry might interact with histone complexes, which have even-numbered symmetry. The highly thermostable N-terminal domain of NUP alone is also capable of binding histones. Indeed, poly-glutamate (mimicking the C-terminal tail of nucleoplasmin) has also been found to de-condense sperm chromatin, presumably through binding positively charged histones, albeit less efficiently than nucleoplasmin. Nucleoplasmin's many phosphorylated residues, as well as some acidic loops, seem to play a part in binding positively charged histones. It is also involved in the de-condensation of sperm chromatin (reviewed in Ref. Nucleoplasmin (NUP) provides a long-lasting store of histones H2A/H2B in Xenopus eggs, which is sufficient for a dozen or so rounds of DNA replication after fertilisation. In particular, histone chaperones prevent potentially toxic histone proteins from binding DNA, a role that is exemplified by the amphibian nucleoplasmin protein. 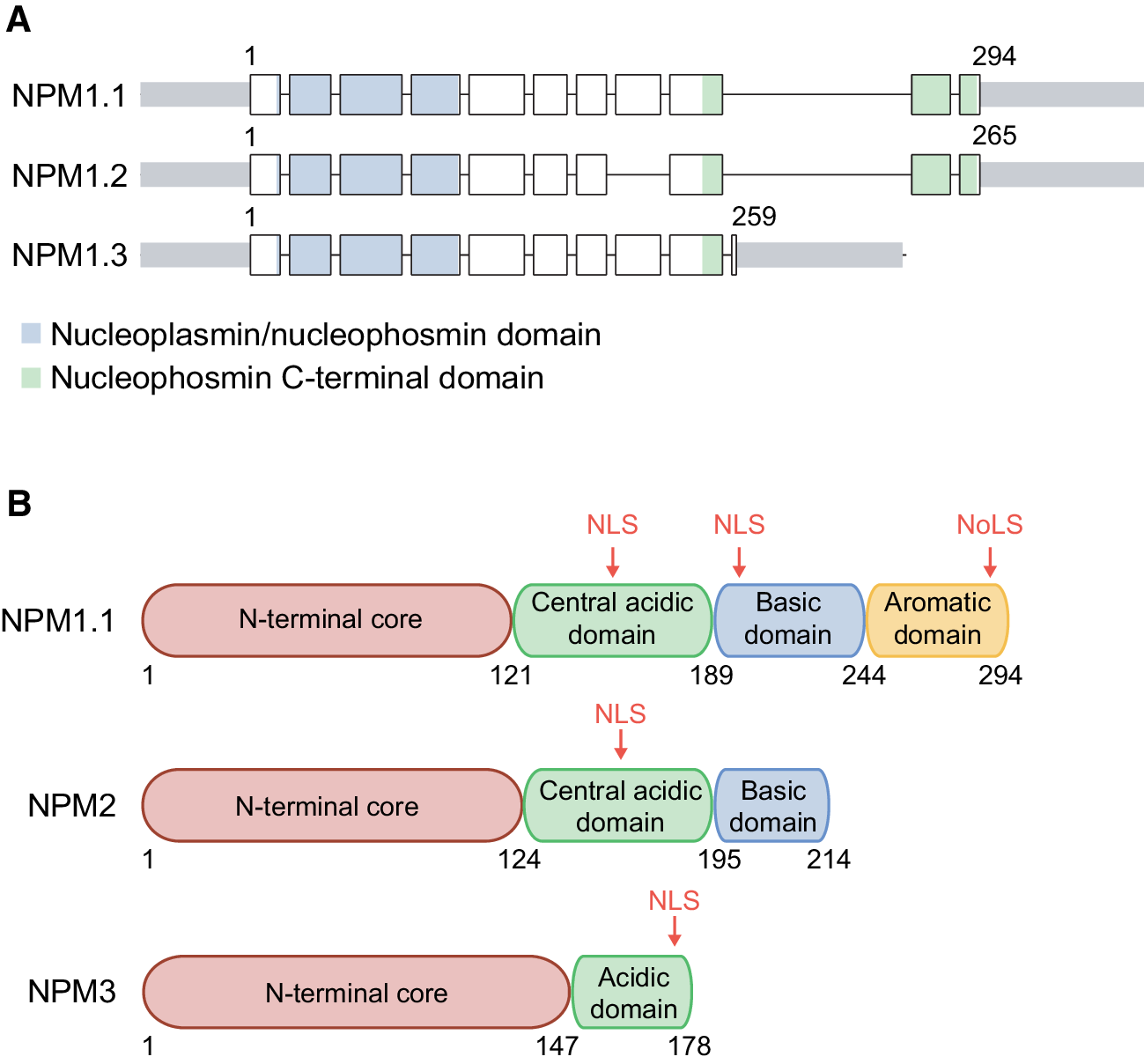
They either temporarily bind and present histones to histone modifying enzymes or hold histones in an inactive state after they are synthesised or released from DNA during transcription, replication and repair.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
